|
The lessons learned in this post will continue through the study of chemical reactions and equations. So, 1 mol contains 6.022×10 23 elementary entities of the substance. We also learned how to calculate the molar mass of a compound using the periodic table and how to convert mass into moles. We learned about Avogadro’s number and how it relates to the mole, which is a unit used to express the amount of a substance. In conclusion, understanding molar mass is an essential concept in chemistry as it allows us to relate the amount of a substance to the number of particles present in it. Specifically, the number is defined as 6.022 \times 10^. Example 3: Finding the Mass of a Number of MoleculesĪvogadro’s number is a fundamental constant that represents the number of particles (atoms, molecules, ions) in one mole of a substance.Add them together: add the results from step 3 to get the total molar mass of the compound. Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound. How to Find Molar Mass Using the Periodic Table The atomic mass is usually found on the periodic table and is given in atomic mass units (amu).The Mole and its Relationship to Avogadro’s Number.Thus, the formula mass of calcium hydrogen carbonate is 117.10 amu and the molar mass of calcium hydrogen carbonate is 117.10 grams per mole (g/mol). However when talking about a mole of an ionic compound we will still use the term molar mass. This is because there are no individual molecules in ionic compounds. For compounds that are not molecular (ionic compounds), it is improper to use the term “molecular mass” and “ formula mass” is generally substituted. The molar mass of the N 2 molecule is therefore 28.02 g/mol. This is referred to as the molecular mass and the molecular mass of any molecule is simply the sum of the atomic masses of all of the elements in that molecule. For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) = 28.02 amu. For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. 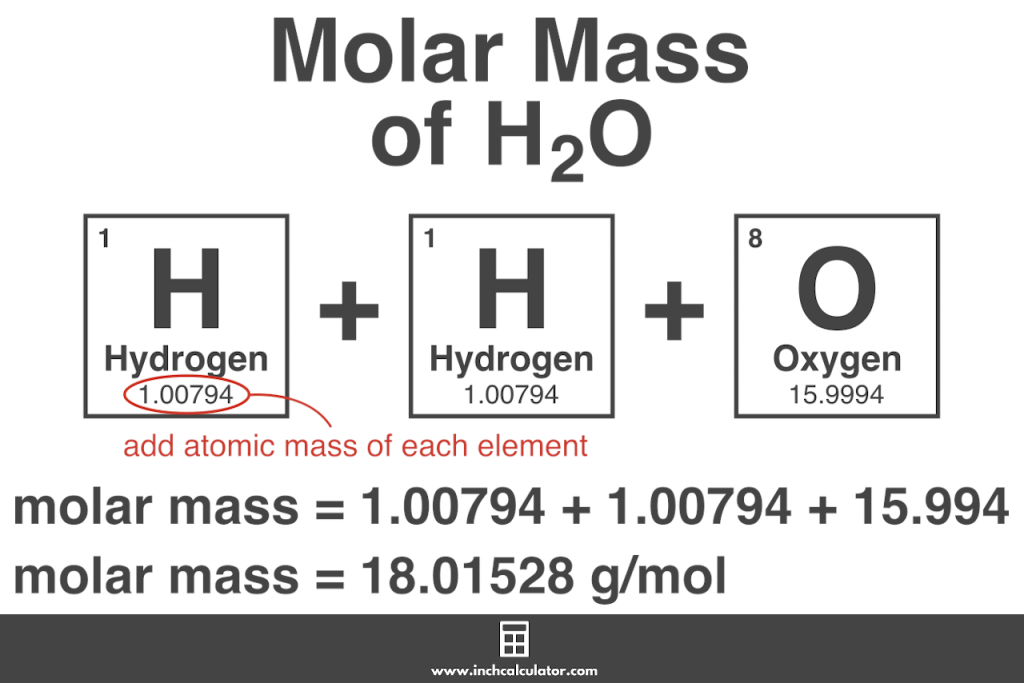
The concept of molar mass can also be applied to compounds. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12. Further, if you have 16.00 grams of oxygen atoms, you know from the definition of a mole that your sample contains 6.022 x 10 23 oxygen atoms. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. For example, the atomic mass of an oxygen atom is 16.00 amu that means the molar mass of an oxygen atom is 16.00 g/mol. Generalizing this definition, the molar mass of any substance in grams per mole is numerically equal to the mass of that substance expressed in atomic mass units. By definition, the molar mass of carbon-12 is numerically the same, and is therefore exactly 12 grams. The relative atomic mass indicates how many times larger the mass of a given atom is than 1/12 the mass of the 12 C carbon isotope. Fe4 Fe (CN)63, NaHCO3, ch3coonh4, h2so4, pb (c2h3o2)23h2o, caso41/2h2o) and press Enter or click Calculate button. Molar mass is typically expressed in either gram ( g) or kilograms (kg). Enter a chemical formula to calculate its molar mass (e.g. Multiply the subscript (number of atoms) times that element’s atomic mass and add the masses of all the elements in the molecule to obtain the molecular mass. We have defined a mole based on the isotopic atomic mass of carbon-12. Find the atomic mass for each element using the mass shown in the Periodic Table or Atomic Weight Table. The colourless stones, when cut, resemble diamonds. The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights (), compiled as part of the 2021 Table of Standard Atomic Weights 2021. Cubic zirconia (zirconium oxide) is a synthetic gemstone. The element was first discovered in this form, resulting in its name. The atomic mass of an element is the relative average of all of the naturally occurring isotopes of that element and atomic mass is the number that appears in the periodic table. Zircon is a natural semi-precious gemstone found in a variety of colours. 
) a carbon atom with six proton and six neutrons in its’ nucleus, surrounded by six electrons. A mole is defined as exactly 6.02214076 × 10 23 particles, e.g., atoms, molecules, ions or electrons.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
